Mammals have evolved flight many times, and we are finally learning how

While birds are the undisputed champions of the sky, having mastered flight during the Jurassic, mammals have actually evolved flight more often than birds. In fact, as many as seven different groups of mammals living today have taken to the air independently of each other.
These evolutionary experiments happened in animals scattered all across the mammalian family tree – including flying squirrels, marsupial possums and the colugo (cousin of the primates). But they all have something in common. It’s a special skin structure between their limbs called a patagium, or flight membrane.
The fact these similar structures have arisen so many times (a process called convergent evolution) hints that the genetic underpinnings of patagia might predate flight. Indeed, they could be shared by all mammals, even those living on the ground.
If this is true, studying patagia can help us to better understand the incredible adaptability of mammals. We might also discover previously unknown aspects of human genetics.
A deceptively simple membrane
Despite being seemingly simple skin structures, patagia contain several tissues, including hair, a rich array of touch-sensitive neurons, connective tissue and even thin sheets of muscle. But in the earliest stages of formation, these membranes are dominated by the two main layers of the skin: the inner dermis and outer epidermis.
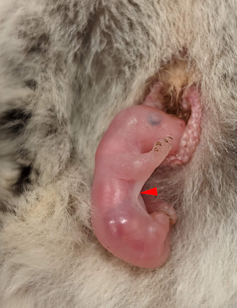
At first, they hardly differ from neighbouring skin. But at some point, the skin on the animal’s sides starts to rapidly change, or differentiate. The dermis undergoes a process called condensation, where cells bunch up and the tissue becomes very dense. Meanwhile, the epidermis thickens in a process called hyperplasia.
In some mammals, this differentiation happens when they are still an embryo in the uterus. Incredibly though, in our main model species – the marsupial sugar glider (Petaurus breviceps) – this process begins after birth, while they are in the mother’s pouch. This provides us with an incredible window into patagium formation.
Starting with the sugar glider, we examined the behaviours of thousands of genes active during the early development of the patagium, to try and figure out how this chain of events is kicked off.
From gliders to bats
We discovered that levels of a gene called Wnt5a are strongly correlated with the onset of those early skin changes – condensation and hyperplasia. Through a series of experiments involving cultured skin tissues and genetically engineered laboratory mice, we showed that adding extra Wnt5a was all it took to drive both of these early hallmarks of patagium formation.
Interestingly, when we extended our work to bats, we found extremely similar patterns of Wnt5a activity in their developing lateral patagia to that in sugar gliders. This was surprising, since bats (placental mammals) last shared a common ancestor with the marsupial sugar glider around 160 million years ago.
Perhaps even more remarkably, we found a nearly identical pattern in the outer ear (or pinna) of lab mice. The pinna is a nearly universal trait among mammals, including innumerable species with no flying ancestry.

A molecular toolkit
Together, these results suggest something profound. Wnt5a’s role in ushering in the skin changes needed for a patagium likely evolved long before the first mammal ever took to the air.
Originally, the gene had nothing to do with flight, instead contributing to the development of seemingly unrelated traits. But because of shared ancestry, most living mammals today inherited this Wnt5a-driven program. When species like gliders and bats started on their separate journeys into the air, they did so with a common “molecular toolkit”.
Not only that, but this same toolkit is likely present in humans and working in ways we don’t fully understand yet.
There are definite limits to our recent work. First, we haven’t made a flying mouse. This may sound like a joke, but demonstrates we still don’t fully understand how a region of dense, thick skin becomes a thin and wide flight membrane. Many more genes with unknown roles are bound to be involved.
Second, while we’ve shown a cause-and-effect relationship between Wnt5a and patagium skin differentiation, we don’t know precisely how Wnt5a does it. Moving forward, we hope to fill in these gaps by broadening the horizons of our cross-species comparisons and by conducting more in-depth molecular studies on patagium formation in sugar gliders.
For now though, our study presents an exciting new view of flight in mammals. We may not be the strongest fliers, but trying is in our DNA.
Charles Feigin, Postdoctoral Fellow in Genomics and Evolution, The University of Melbourne
This article is republished from The Conversation under a Creative Commons license. Read the original article.








